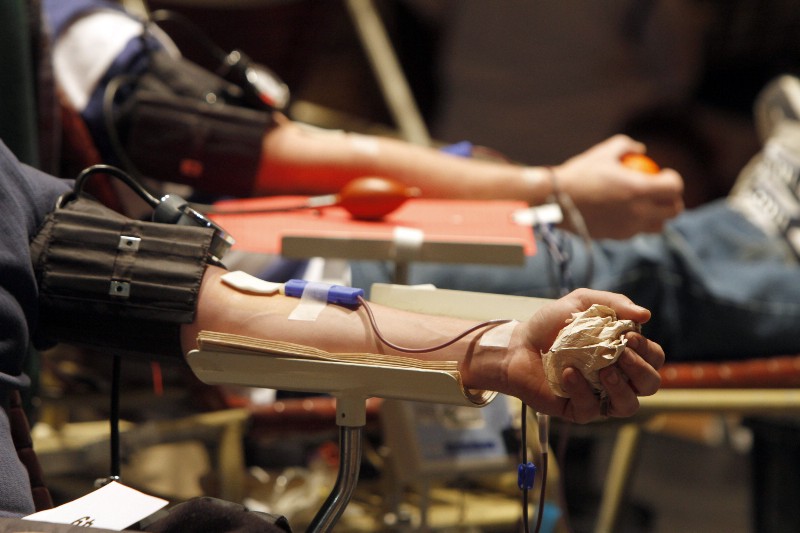
The Food and Drug Administration (FDA) released new guidance for the country’s blood banks on Friday urging them to screen all blood for the Zika virus.
The new recommendations extend to all states, even those without any cases of Zika cropping up. The FDA noted that it thinks there is a risk of spreading the virus through blood transfusions.
To screen blood, hospitals and blood banks were urged to use one of two experimental tests, called nucleic acid tests, that have only been provisionally approved by the FDA for this purpose on an investigational basis. The other option is to decontaminate plasma and platelets with pathogen reduction technology.
The speed with which they’re being urged to begin testing could present serious challenges in many places. Eleven states — Alabama, Arizona, California, Georgia, Hawaii, Louisiana, Mississippi, New Mexico, New York, South Carolina and Texas — have been given just four weeks to get screening up and running because they are near areas with locally contracted Zika cases or have an increased likelihood that residents have been traveling to places with high rates of infections. All other states will have 12 weeks.
“This is a bombshell, because this is extremely rapid introduction of a new test nationwide that’s almost unprecedented,” Dr. Jeffrey McCullough, emeritus professor of laboratory medicine and pathology at the University of Minnesota Medical School, told the New York Times. “To try to implement this, in four weeks, is really, really difficult.”
The new testing may end up meaning hospitals have to pay more for each unit of blood, possibly up to $8 more per unit, given the cost of testing the blood for Zika. The FDA’s director of the Center for Biologics Evaluation and Research said he had no information about any financial assistance there might be to deal with the cost of implementation.
Experts told the Times that the FDA’s move is a way to deal with the virus while Congress fails to provide funding for a more robust response. President Obama has requested $1.9 billion in emergency funding to fight the virus, but lawmakers left for their summer break without passing a package thanks to Republican opposition.
Without federal funding, local health departments haven’t been able to screen patients for the virus in their blood or urine. “In some ways the inaction from Congress has forced the FDA to adopt this position,” Dr. Peter J. Hotez, the dean of the National School of Tropical Medicine at Baylor College of Medicine, told the Times. “They have no other choice.”
There have already been more than 1,600 confirmed cases of Zika in the continental United States, including some that were contracted from local mosquitoes and not travel. Those cases have been concentrated in Florida, where blood banks have already been testing for Zika, and testing has been going on in Puerto Rico since March. The Centers for Disease Control found a surprisingly high share of blood donors in Puerto Rico showed signs of being infected with Zika.